The producer of an award-winning portable dental x-ray device has moved all injection molding to the United States from China to improve communications as it rapidly expands output.
“We had started out molding off shore in China, but due to the logistics, communication concerns and the time delay to respond to mold changes or repairs, we have moved all molding to the U.S. for convenience to meet deadlines and eliminate the shipping cost of overseas operations,” Gar Hendry, manufacturing engineering manager for Aribex (Orem, UT), told PlasticsToday.
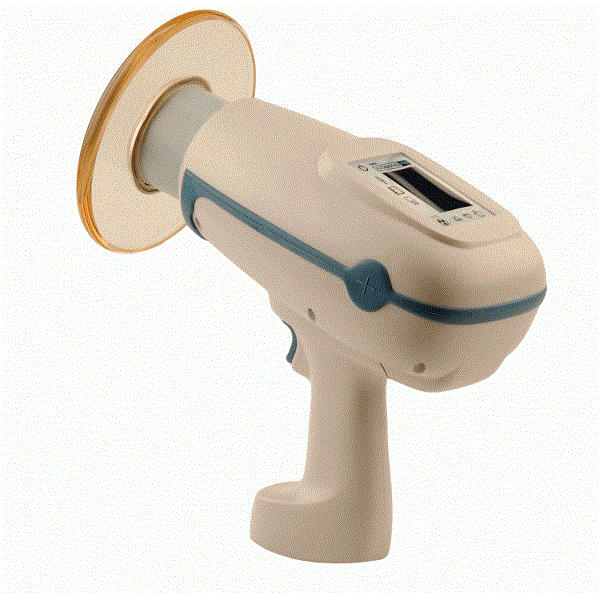
Aribex makes the NOMAD Pro handheld x-ray system that can be used in or out of a dental office. The rechargeable device is about the size of a battery-operated power drill. In a big advance, it is internally shielded so that a technician can take x-rays while standing or sitting next to the patient.
No more lead walls
In traditional systems, technicians stand behind shields away from the x-ray system, which is usually wall-mounted and sometimes located in a special room. Patients receive low exposure to radiation in the NOMAD Pro as a result of other technical improvements.
NOMAD was invented by Clark Turner, a technical expert in x-rays, who was once told by his personal dentist that he’d like an x-ray unit to take on humanitarian trips to third world countries. The first NOMAD was commercially introduced in 2007.
“Our original customers were dentists who were going on humanitarian trips and planning to use it out of the office,” said Turner, “but once they started using it they said this would be really cool to have in the office instead of the regular wall-mounted systems.”
Improved productivity
The NOMAD Pro was introduced in 2008, 30% lighter than the original device and featuring advanced lithium-polymer battery technology. According to Turner, one unit can take the place of three to four wall-mounted x-rays due to its portability.
“At the beginning of the product design we were concerned that with any of the plastics we had to choose from it had to meet several requirements for safety, product durability in the field, ease of cleaning and chemical resistance,” Hendry told PlasticsToday. “These requirements narrowed the choices down to the polycarbonate family of plastics. After looking at the available options we decided on Lexan 9112. It met the safety requirements for flame resistance, electrical isolation, impact resistance, chemical resistance and was available in multiple color choices.”
Aribex chose filled thermoplastic polyurethanes to assist in blocking the x-ray beam. “The Lexan and TPU had the qualities to last in the field and meet all federal and international regulations,” Hendry said. Aribex, which is now using contract injection molders in the United States, employs more than 50 people.
The NOMAD Pro is a silver award winner in the 2012 Medical Device Excellence Award (MDEA) competition. Winners were announced last month in Philadelphia at MD&M East. The entry was submitted by IQMS (Paso Robles, CA).
Output will double
There are nearly 10,000 NOMAD devices in service around the world. The move to domestic production was spurred by Turner’s ambitions expansion plans. “I really plan to build and grow this company,” said Turner. “I expect to double the size over the next three to four years through taking our products to international markets and creating new ones for medical and industrial industries. The dentists are expressing a need. I aim to meet it.”
Turner is targeting veterinary, medical, industrial, and security applications as well as expanded penetration in the dental market.