Mobility and speed are significant trends in new medical devices and equipment. ABS, polycarbonate and other structural plastics play significant roles because of their light weight and portability.
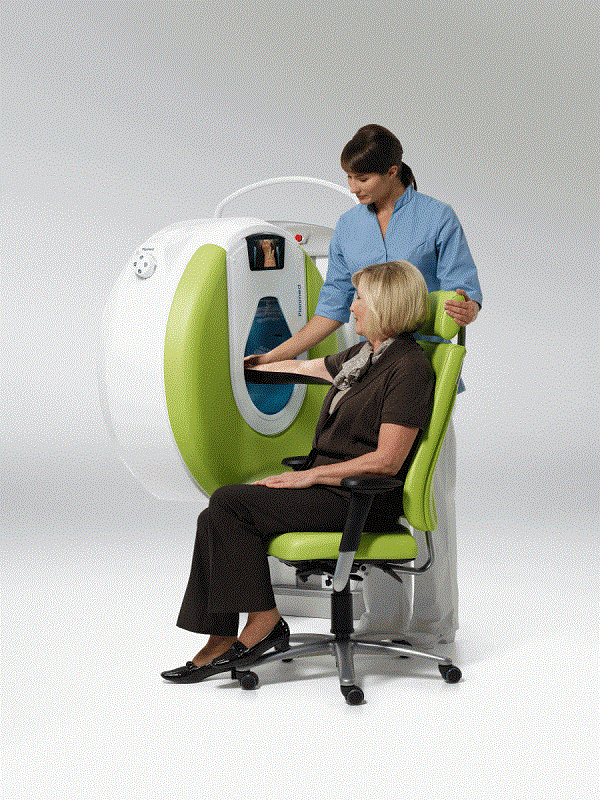
One example is the Planmed Verity Extremity Scanner, a new, mobile CT scanner for orthopedic imaging of
the extremities that was the best-in-show winner in this year’s Medical Design Excellence Awards (MDEA) competition. Winners were announced last month during MD&M East in Philadelphia.
The Planmed Verity is a low dose imaging system that provides fast 3D imaging of peripheral skeletal fractures and disorders at the point-of-care.
A research collaboration between the Massachusetts General Hospital (MGH, Boston, MA) and Finnish Planmed Oy is now studying the benefits and value of 3D imaging options of cone beam computed tomography (CBCT) technology for imaging of extremities.
Medical staff at MGH is studying the image quality, speed of examination, and accuracy of diagnosis versus traditional CT equipment.
“Planmed is actively involved in a wide variety of important research projects concerning both of our product lines: mammography and breast cancer detection as well as orthopedic imaging,” says Vesa Mattila, VP, Planmed Oy. Mattila says the equipment is designed to find subtle extremity fractures at the first visit to the clinic–fractures that may have been missed using only 2D radiographs.
Images are acquired using a short X-ray pulse instead of continuous radiation, enabling a low radiation dose.
In another example (Gold MDEA winner),a device called the King Vision video laryngoscope uses cell phone camera and digital display technology to simulate traditional laryngoscopes at a much lower cost than video laryngoscopes. The purpose of the invention is to allow tracheal intubation inside and outside the hospital.
CARDIOHELP (silver award) is described as the world’s smallest portable heart-lung support system providing life support to replace or support a patient’s circulation and respiration. MAQUET Cardiovascular last year received U.S. Food and Drug Administration 510(k) clearance to market CARDIOHELP.
“Cardiovascular disease is the leading cause of death globally, with an estimated 17.5 million deaths each year,” said Jeremy Cannon, M.D., trauma surgeon at Brooke Army Medical Center in San Antonio, Texas. “Many of these individuals experience cardiogenic shock because vital organs are not adequately supplied with oxygen. By quickly connecting a patient to a cardiopulmonary support system, the CARDIOHELP System will allow clinicians to gain valuable time that could save the patient’s life.”
It’s light enough to be carried by one person and compact enough to be transported in a helicopter or vehicle.
Source : http://www.plasticstoday.com/articles/medical-devices-emphasize-speed-mobility0611201201