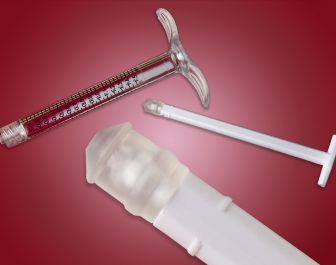
At the Medical Design and Manufacturing (MD&M) East exposition, Teknor Apex Company will introduce a series of thermoplastic vulcanizate (TPV) elastomers spanning a wide range of hardness levels exhibits high purity and resilience for replacing rubber in medical applications and provides performance and processing advantages over other TPEs.
Medalist MD-200 Series elastomers range from ultra-soft 15 Shore A compounds to 80 Shore A grades, avoiding need for pre-drying because they are non-hygroscopic, and have a light natural color that permits efficient use of colorants. According to the release, all grades have been certified to pass ISO 10993-5 and are Drug Master File listed with the US Food and Drug Administration (FDA). The compounds do not contain extractable heavy metals commonly used in the curing of thermoset rubber. Because they exhibit lower oxygen absorption than non-vulcanizate TPEs, pharmaceutical contents are properly protected when used in seals or gaskets for vials.
“The TPV technology for the MD-200 series is one of multiple polymer chemistries employed by Teknor Apex in developing our Medalist family of medical elastomers,” said Keith Saunders, Senior Market Manager for the Thermoplastic Elastomer Division. “We uniquely manufacture our TPV compounds ‘from scratch’, starting with polymers and all other basic ingredients, rather than using a masterbatch intermediary.”
According to Teknor Apex, as TPVs, Medalist MD-200 compounds exhibit greater resilience than other TPEs, perform well in high-temperature end uses, and are more chemical-resistant. They can be sterilized in steam, autoclave, or gamma irradiation processes. Like all thermoplastics, they process more easily and quickly than rubber, offer greater design freedom, and are recyclable. Most grades in the series adhere well to PP in over-molding.
Used commercially as a replacement for thermoset rubber in syringe stoppers, Medalist MD-200 compounds have been shown to meet the requirements of ISO 7886 for single-use hypodermic syringes and exhibit a low coefficient of friction for consistent travel force in glass and PP barrels, the company added.
The MD&M East exposition will be held from May 22-24 in Philadelphia, the US. Alongside the exposition, the MD&M East conference is taking place from today (May 21) to May 24.
Source : http://adsalecprj.com/Publicity/MarketNews/lang-eng/article-123416/Article.aspx